Journal of Ocular Biology
Download PDF
Research Article
Treatment of Staphylococcus Aureus with Antibiotic and Quorum-Sensing Inhibitor Combinations Reduces Severity of Keratitis
Mark DP Willcox1*, Emma BH Hume1, Tracey L. Schubert1,2 and Naresh Kumar3
- 1School of Optometry and Vision Science, University of New South Wales, Australia
- 2Vision CRC, Sydney, Australia
- 3School of Chemistry, University of New South Wales, Australia
Address for Correspondence: Mark DP Willcox, School of Optometry and Vision Science, University of New South Wales, NSW 2052, Australia, Tel: 61 2 9385 4164; Fax: 61 2 9313 6243; E-mail: m.willcox@unsw.edu.au
Citation: Willcox MDP, Hume EBH, Schubert TL, Kumar N. Treatment of Staphylococcus Aureus with Antibiotic and Quorum-Sensing Inhibitor Combinations Reduces Severity of Keratitis. J Ocular Biol. 2017;5(1): 5.
Journal of Ocular Biology | ISSN: 2334-2838 | Volume: 5, Issue: 1
Submission: 13 October, 2017| Accepted: 31 October, 2017 | Published: 15 November, 2017
Submission: 13 October, 2017| Accepted: 31 October, 2017 | Published: 15 November, 2017
Copyright: © 2017 Willcox MDP, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Purpose: To examine whether treatment with ciprofloxacin and fimbrolides improves the outcome of Staphylococcus aureus keratitis.
Methods: A S. aureus strain was tested for the minimum inhibitory concentration (MIC) of ciprofloxacin and fimbrolides, and whether they had synergistic antimicrobial activity. The effect of fimbrolides on production of α-toxin was measured after growth at sub-MIC and testing supernatants for hemolysis. Cytotoxicity of the fimbrolides was measured by adding increasing concentrations to corneal epithelial cells and measuring release of lactate dehydrogenase. Ten hours after scratching corneas of mice and applying bacteria, treatment was initiated with vehicle, ciprofloxacin, fimbrolide, or a combination of ciprofloxacin and fimbrolide. The number of infecting S. aureus cells was measured after 6 hours treatment, and clinical responses were recorded (maximum score 20).
Results: The MIC of the fimbrolides was 6.25-12.5 μM and growth in fimbrolides reduced production of α-toxin by > 90%. Fimbrolides were only cytotoxic at > 25 μM. Treatment of keratitis with ciprofloxacin significantly (p < 0.05) reduced numbers of S. aureus in corneas. Although there was minimal synergism between the two antimicrobials in vitro and treatment with the fimbrolide/ciprofloxacin combination in vivo did not significantly reduce numbers of S. aureus, there was a significantly reduced clinical score (by > 45%; p < 0.05) of the combination compared to single application of antimicrobials or vehicle control (average 4.25 ± 0.35).
Conclusion: Application of non-toxic concentrations of quorum-sensing inhibitor fimbrolides in combination with ciprofloxacin reduce the numbers infecting bacteria to the same extent as ciprofloxacin alone, but also significantly improved the clinical outcome of S. aureus keratitis. Fimbrolides may be useful adjunct agents to treat keratitis.
Introduction
Staphylococcus is a leading cause of bacterial keratitis in humans [1]. Bacterial keratitis is a major cause of ocular morbidity and avoidable visual impairment worldwide with concomitant potential for increased social and health care costs [2,3]. Treatment with either fortified amino glycoside and cephalosporin or a monotherapy with quinolones has been used for the treatment of Keratitis [4,5]. The pathology associated with S. aureus keratitis might be produced by release of microbial toxins, not simply by replication of bacteria in the cornea [6,7]. A number of S. aureus virulence factors have been implicated in the pathogenesis of corneal disease. These include factors such as α-toxin and β-toxin both of which are membrane active toxins that permeabilize mammalian cell membranes leading to cell death [6-8]. The production of α-toxin and β-toxin is controlled at least in part, by the staphylococcal accessory gene regulator (agr) quorum-sensing system [9].
Agents that disrupt quorum sensing or bacterial cell-to-cell communication are emerging as a potential therapeutic candidate [10-12]. Fimbrolides are a unique class of halogenated furanones produced by the marine red algae Delisea pulchra [13]. Synthetic variants of these naturally occurring compounds have been synthesized and shown to prevent microbial colonisation of surfaces and production of virulence factors [14-18]. These compounds can disrupt the autoinducer-1 (AI-1) quorum-sensing system that is possessed by many Gram-negative bacteria, but they can also disrupt AI-2 (luxS) signaling systems in Gram-positive and Gram-negative bacteria [19-23]. They are similar in structure to the acyl-homoserine lactone (AHL) quorum sensing molecules produced by Pseudomonas aeruginosa, and AHLs down regulate expression of sarA and the accessory gene regulator agr in S. aureus [24]. S. aureus isolates possessing a non-functional agr exhibit reduced pathogenicity in the rabbit eye [8]. Thus, products that inhibit expression of agr or production of its associated virulence factors may reduce disease pathology in animal models of S. aureus keratitis. The present study examined efficacy of two novel fimbrolides to synergize with conventional antibiotics in vitro. Application of fimbrolide compounds alone or in combination with antibiotics was examined in relation to clinical score and number of bacteria recovered in infected corneas in vivo.
Materials and Methods
Bacterial strains
A clinical S. aureus isolate, strain 38, from a corneal ulcer were used in this study [25]. Stock cultures were stored in 30% glycerol at -80 °C prior to use.
Fimbrolide compounds
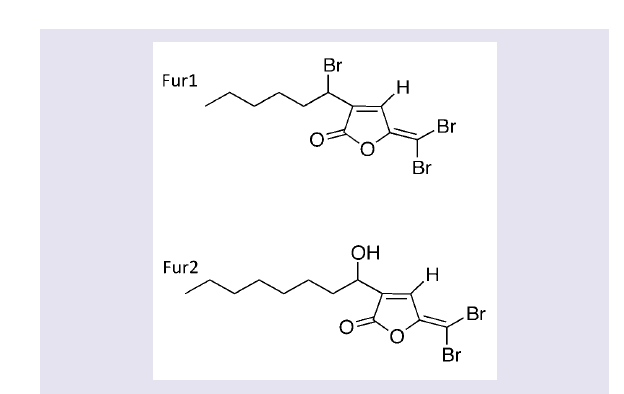
Fimbrolides were synthesized according to published methods (Fur1 and Fur2; (Figure 1) [26,27]. Compounds were purified by chromatography with CH2Cl2/light petroleum as the mobile phase in a 1:1 ratio and were > 95% pure. Compounds were diluted to a stock concentration of 0.5 mM in absolute ethanol (Sigma-Aldrich, Sydney, Australia) prior to use.
Minimal inhibitory broth dilution tests
The minimal inhibitory concentration (MIC) of the fimbrolides and ciprofloxacin was determined. Bacteria were cultured in tryptone soya broth (TSB; Oxoid, Basingstoke UK) overnight at 37 °C and then resuspended in TSB. Two microliters of stock fimbrolide (in absolute ethanol) or ciprofloxacin hydrochloride (ICN Biomedicals Australasia, Australia; diluted to stock concentration of 10 mg/ml in sterile water) was added to a final concentration of 0.31 μM to 50 μM for fimbrolide or 0.002 μg/ml to 1 mg/ml for ciprofloxacin in TSB containing 1 x 105 S. aureus cfu/ml in a chequerboard design [28]. Absolute ethanol (2 μl) in TSB was used as a control. The MIC was determined as the lowest concentration of fimbrolide or ciprofloxacin that inhibited bacterial growth after incubation for 18 hours at 37 °C.
Fractional inhibitory concentration of ciprofloxacin in the presence or absence of fimbrolides
Synergy of the antimicrobials was determined using the sum of the fractional inhibitory concentrations (FIC) of antibiotic and fimbrolide. FIC index was used to determine synergy and was calculated using the following equation: FIC index = ((MIC of antibiotic + fimbrolide)/MIC of fimbrolide)/(MIC of antibiotic + fimbrolide)/MIC of antibiotic)). Synergism was defined as an FIC index of ≤ 0.5 indifference as an FIC index of > 0.5 but ≤ 4 and antagonism as an FIC index of > 4 [29].
Effect of fimbrolide on production of α-toxin
The effect of the fimbrolide on the production of α-toxin was measured Briefly; S. aureus 38 was grown in TSB overnight at 37 °C in the presence or absence of a sub-inhibitory concentration of each furanone (2.5 μM). After growth, cells were removed by centrifugation (5,000 g 10 mins) and the supernatants removed and filtered through 0.22 μm filters. Citrated rabbit erythrocytes were purchased from Bioreclamation IVT (NY, USA), washed in PBS (8 g/l NaCl, 0.2 g/l KCl, 1.15 g/l Na2HPO4, 0.2 g/l KH2PO4, pH 7.3) and resuspended in PBS and added to wells of a 96 well micro titer plate at approximately 107 erythrocytes/well [30]. Bacterial supernatants were added and the mixtures allowed incubating at 37 °C for 1 hour. After that time, plates were centrifuged, the supernatants removed and the level of hemolysis was estimated by measuring the absorbance of the supernatants at OD 545 nm.
Cytotoxicity assay
Prior to experiments with animals, the cytotoxicity of the fimbrolides was determined indirectly by measuring the level of lactate dehydrogenase (LDH) in cell culture supernatants. An immortalized human corneal epithelial (HCE) cell line was cultured in supplemented hormone epithelial medium (SHEM) as previously described [31]. Cells (3.6 x 105/ml) were seeded and cultured in 24-well plates to a confluent monolayer. Confluent cells were incubated in modified Eagle’s media/Hams F12 (MEM/F12; Trace Bioscience, Sydney, Australia) containing 0.6% bovine serum albumin (Sigma, St Louis, USA) for 6 hours [32]. Media was then replaced with SHEM and cells were challenged with concentrations of fimbrolide (12.5 μM to 25 μM from the stock ethanol solutions) in media for 24 hours at 37 °C. Supernatants were removed and the level of LDH in cell supernatant was measured using a Cytotox 96 ® non-radioactive cytotoxicity assay procedure (Promega, Madison, WA, USA) according to the manufacturer’s instructions. Controls included HCE incubated in equivalent quantity of absolute ethanol (fimbrolide media) in SHEM(0% cytotoxicity), and HCE incubated at 37 °C for 45 min in 10 μl lysis solution (Promega) and 90 μl SHEM to give 100% lysis of cells (i.e. 100% cytotoxicity). A cytotoxic concentration of fimbrolide was defined as a concentration which caused the release of > 30% the maximum LDH.
Formation of aqueous solutions of fimbrolides and ciprofloxacin
Due to the relative hydrophobicity of the fimbrolides, the fimbrolides and ciprofloxacin were dissolved in 2-hydroxypropyl-β-cyclodextrin (28 mg/ml; Sigma-Aldrich, St Louis, MO, USA) and sodium acetate (0.01 M) in 0.8% (w/v) NaCl prior to being tested in vivo, as we were concerned with adding an ethanol containing solution (i.e. dilutions of the stock fimbrolides) directly to eyes. The antimicrobial activity after dissolving in the cyclodextrin/acetate was determined by applying 5 μl drops on to Sensitest agar (Oxoid, Basingstoke, UK) pre-coated with 1 colony of S. aureus 38 suspended in 0.9% sodium chloride. These plates were then incubated at 37 °C for 24 h and zones of inhibition of bacterial growth determined. Controls of antimicrobial alone and vehicle alone were also examined. To ensure that the cyclodextrin vehicle was not toxic to the mouse cornea, ten microliters of cyclodextrin/sodium acetate was applied to the right eye of A/J mice hourly for 6 h. The left eye of each animal served as a control and was not treated. Following the 6 hourly treatments corneas were stained with fluorescein and mice were monitored with slitlamp as described below. Inbred A/J mice were obtained from the Animal Resource Centre, Perth, Australia. Mice were maintained according to the guidelines put forth in the Association for Research in Vision and Ophthalmology (ARVO) Statement for the use of animals in ophthalmic and vision research. Institutional ethics clearance was obtained prior to initiation of any animal experiments.
Corneal infection of mice
Inbred 6-8 week old mice were anaesthetized with an intraperitoneal injection of Avertin (2,2,2 tribromoethanol; 125 mg/kg) and the analgesic buprenorphine 0.1 mg/kg was administered subcutaneously. Corneas of both eyes were incised in a cross-shaped pattern with a sterile 27 gauge needle. Ten micro liters of 0.6% acetyl cysteine (Sigma, St Louis, MO, USA) was applied onto the cornea to break up the tear film, and then removed with a sterile cotton swab. Bacteria were grown for 12 hours in tryptone soya broth (TSB; Oxoid, Basingstoke UK) at 37 °C. Bacteria were concentrated by centrifugation (3000 g) in fresh TSB to approximately 4 x 1010 cfu/ml. Ten microliters of bacterial suspension of strain S. aureus 38 was applied directly onto the wounded cornea. The left eye of each animal served as a control and was scratched but not infected. A minimum of 15 animals was included in each treatment group.
Mouse eyes were treated hourly for six hours starting 10 hours post-bacterial infection with either vehicle alone (hydroxypropyl-β-cyclodextrin and sodium acetate), or 0.3% ciprofloxacin, or 12.5 μM of either fimbrolide alone or a combination of ciprofloxacin or either fimbrolide (0.3% ciprofloxacin and 12.5 μM fimbrolide) in cyclodextrin/acetate [33]. Mice were monitored using a slit-lamp by a masked observer at 16 hours post bacterial infection as previously described [34]. Each of five parameters (exudate, epithelial defect, corneal infiltrate extent, corneal opacity and corneal edema) was graded on a scale of 0 (none) to 4 (severe) in 0.1 increments during slit lamp examination. Summation of the parameter grades produced a single slit-lamp examination score which could range from 0 (normal eye) to a theoretical maximum of 20. After euthanasia, corneas were collected 16 hours post-infection and bacteria enumerated as described previously in homogenized corneas [25]. Results were expressed as mean cfu/ml ± stand error of the mean (SEM).
Statistical analysis
Corneal pathology and bacterial numbers were expressed as the mean ± SEM. The variables were compared between treatment groups using one-way analysis of variance (ANOVA). Variables that were significantly between treatments were further analyzed using Browne-Forsythe’s T3 correction.
Results
The MIC for ciprofloxacin was 0.5 μg/ml, for Fur1 was 6.25 μM and for Fur2 was 12.5 μM. There was no antibacterial synergistic activity of Fur1 with ciprofloxacin, but there was synergy (ΣFIC 0.5) with Fur2 and ciprofloxacin. Growth in the presence of either fimbrolide resulted in inhibition by > 90% of the production of α-toxin. The fimbrolide at the sub-MIC of concentration had no effect of bacterial growth (data not shown).
Cytotoxicity of the fimbrolides was determined indirectly by measuring the level of LDH in cell culture supernatant. Fur1 and Fur2 exhibited cytotoxicity at ≥ 25 μM. There was a small effect on antimicrobial activity of dissolving the fimbrolides in cyclodextrin/sodium acetate solution; the zone of inhibition on the agar plate changed from 5.5 mm to 5.0 mm. Previous studies have shown that cyclodextrins alone do not affect α-toxin production [30]. There were no clinical changes in the mouse cornea or conjunctiva after repeated instillation of the cyclodextrin/sodium acetate solution.
Effect of fimbrolide treatment on eye infected with S. aureus
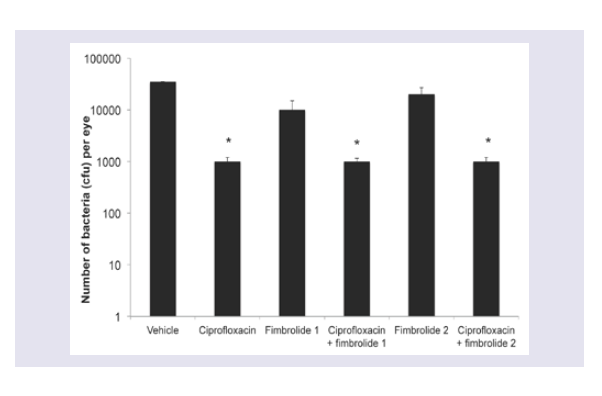
Treatment with ciprofloxacin alone for 6 h reduced the numbers of S. aureus 38 recovered by 94 % (Figure 2; p ≤ 0.05). There was no further reduction in bacterial numbers when combined with either fimbrolide, but the combinations retained the 94% reduction in bacterial numbers demonstrating there was no antagonism. There was no reduction in bacterial numbers with either fimbrolide alone.
Figure 2: Numbers of bacteria recovered from the corneas of A/J mice infected with S. aureus following 6 h of treatment with fimbrolide and/or ciprofloxacin. *indicates significant differences between treatments and respective vehicle controls (p ≤ 0.05).
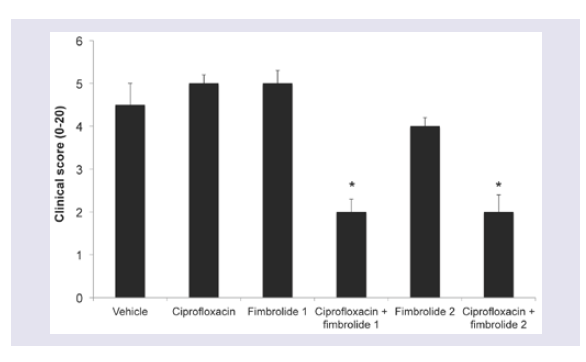
The scratch control non-infected corneas had clinical scores (sum of scores for exudate, epithelial defect, corneal infiltrate extent, corneal opacity and corneal edema-maximum score = 20) of 1.1 ± 0.2. Corneas infected with S. aureus had higher clinical scores than scratch controls (p < 0.05).Treatment with ciprofloxacin or Fur1 or Fur2 alone did not reduce the clinical score (Figure 3). However when ciprofloxacin was combined with Fur1or Fur2 the clinical score was significantly reduced compared to treatment with the vehicle, fimbrolide alone or ciprofloxacin alone (Figure 3; p ≤ 0.05).
Figure 3: Clinical score of A/J mouse eyes infected with S. aureus following 6 h treatment with fimbrolide and/or ciprofloxacin. *indicates significant differences between treatments and respective vehicle controls (p ≤ 0.05).
Examining the different aspects of the clinical score, there were additional differences between treatments for the scores for corneal infiltrates or corneal opacity (0-4 scales). Treatment with the ciprofloxacin/Fur1 or ciprofloxacin/Fur2 combinations resulted in significant decreases (p ≤ 0.05) in corneal opacity compared to vehicle (0.5 ± 0.1 or 0.6 ± 0.1 vs. 1.5 ± 0.2 respectively) or compared to ciprofloxacin treatment alone (vs. 1.3 ± 0.2). Similarly treatment with the ciprofloxacin/Fur1 or ciprofloxacin/Fur2 combinations resulted in significant decreases (p ≤ 0.05) in corneal infiltrates compared to vehicle (1.2 ± 0.1 or 1.2 ± 0.1 vs. 2.1 ± 0.2 respectively) or compared to ciprofloxacin treatment alone (vs. 2.3 ± 0.1).
Discussion
The purpose of this study was to determine whether fimbrolides can synergize with ciprofloxacin in vitro and in an animal keratitis model as an indication of the capacity of fimbrolides to improve clinical outcomes of staphylococcal keratitis. Fimbrolides (also known as furanones) have been shown to inhibit the growth of Grampositive bacteria in vitro and similar compounds have shown potent antimicrobial activity against S. aureus [35-37].
This study demonstrated that fimbrolides prevented the growth of S. aureus with MICs of between 6.25-12.5 μM. Fimbrolides have previously been reported to inhibit the growth of S. aureus 35 and AHLs, which are structurally related to the fimbrolides, can also inhibit S. aureus growth, albeit at higher MICs of 30 μM or above [24,35]. Fur2 produced a synergistic antibacterial effect with S. aureus in vitro [38]. The fimbrolides also significantly reduced the production of agr-regulated α-toxin. AHLs have been shown to inhibit the expression of agr as well as sacA, genes that globally regulate production of α-toxin along with other virulence factors of S. aureus [24]. An AHL containing a C12 acyl side chain was effective but ones containing smaller side chains were ineffective, and 3-oxo substitution on the side chain was critical for activity [24]. Subsequently, 3-acyltetramic acids or 3-acyltetronic acids (of similar molecular structure to fimbrolides) have been shown to inhibit growth of S. aureus (MICs of≥ 12.5 μM) and inhibit of the expression of agr. Thus it seems likely that the inhibition of α-toxin production by the fimbrolides in the current study was as a consequence of inhibition of the expression of agr.
There was a significant decrease in bacterial numbers upon antibiotic treatment with ciprofloxacin alone. This data is very similar to that of Dajcs JJ, et al. [33]. Who found a reduction in bacterial numbers with ciprofloxacin sensitive strains of S. aureus, even though the two animal models differed with respect to animal type (mouse vs. rabbit) and application of bacteria (topical vs. corneal injection). Dual therapy with the ciprofloxacin/fimbrolide combinations did not further decrease the numbers of bacteria despite a synergistic effect between Fur2 and ciprofloxacin in vitro.
Interestingly, the clinical outcomes did not follow the bacterial findings in vivo. The combination of ciprofloxacin and fimbrolides produced the most significant reductions in overall clinical scores, corneal opacity and corneal infiltration, yet the combination did not reduce bacterial numbers more than monotherapy with ciprofloxacin. This might mean that the reduction in bacterial numbers, to 1000 colony forming units per cornea, given by one day administration of ciprofloxacin was not sufficient to improve clinical score. This is perhaps not surprising when one considers that the administration of topical antibiotic for human keratitis is as frequent as hourly during the first 24-48 hours to improve clinical outcome [38,39]. However, co-administration of ciprofloxacin with either fimbrolide reduced the clinical response within 24 hours.
The reduction in production of α-toxin by the fimbrolides probably influenced the clinical scores as these is known to be important virulence factor in the cornea, and inhibition of α-toxin production by cholesterol in cyclodextrin has been shown to reduce corneal pathology [6-8,40]. Indeed, strains unable to produce α-toxin replicate in corneas to the same amount as α-toxin producing strains, but the clinical scores are much reduced [6]. Similarly, administration of neutralizing antibodies to α-toxin reduces clinical score without affecting numbers of infecting S. aureus [7]. In the current study, reduction in α-toxin production was not sufficient to reduce clinical score, as treatment with fimbrolide alone had no significant effect.
A potential factor in the clinical response is the finding that AHLs can modulate host immune and inflammatory responses [41,42]. Whether fimbrolides also have this activity is not currently known, but may be they have had an impact on aspects of the clinical response if there is any such anti-inflammatory effect. The present study did not determine the number of neutrophils in corneal tissue, but it is known that this is important in the disease process and it is something that should be assessed in future studies [25,43,44].
and In the present study, fimbrolides inhibited toxin production and combination with ciprofloxacin reduced the corneal pathology during infection. Although the fimbrolide/ciprofloxacin combination did not provide any added benefit in terms of numbers of infecting bacteria, the combination reduced the numbers of infecting bacteria to the same extent as ciprofloxacin alone. Thus fimbrolides may be useful as an adjunct therapy for treating S. aureus keratitis.
Declaration of interest: Two of the authors (Naresh Kumar and Mark DP Willcox) have patents on the synthesis and action of fimbrolides as quorum-sensing inhibitors. There are no other declarations of interest.
References
- Asbell P, Stenson S (1982) Ulcerative keratitis. Survey of 30 years' laboratory experience. Arch Ophthalmol 100: 77-80.
- Keay L, Edwards K, Naduvilath T, Forde K, Stapleton F (2006) Factors affecting the morbidity of contact lens-related microbial keratitis: a population study. Invest Ophthalmol Vis Sci 47: 4302-4308.
- Schaefer F, Bruttin O, Zografos L, Guex-Crosier Y (2001) Bacterial keratitis: a prospective clinical and microbiological study. Br J Ophthalmol 85: 842-847.
- McAllum PJ, McGhee CN (2003) Prescribing trends in infectious keratitis: a survey of New Zealand ophthalmologists. Clin Exp Ophthalmol 31: 496-504.
- Baum J, Barza M (2000) The evolution of antibiotic therapy for bacterial conjunctivitis and keratitis: 1970-2000. Cornea 19: 659-672.
- Callegan MC, Engel LS, Hill JM, O'Callaghan RJ (1994) Corneal virulence of Staphylococcus aureus: roles of alpha-toxin and protein A in pathogenesis. Infect Immun 62: 2478-2482.
- Hume EB, Dajcs JJ, Moreau JM, O'Callaghan RJ (2000) Immunization with alpha-toxin toxoid protects the cornea against tissue damage during experimental Staphylococcus aureus keratitis. Infect Immun 68: 6052-6055.
- O'Callaghan RJ, Callegan MC, Moreau JM, Green LC, Foster TJ, et al. (1997) Specific roles of alpha-toxin and beta-toxin during Staphylococcus aureus corneal infection. Infect Immun 65: 1571-1578.
- Yarwood JM, Schlievert PM (2003) Quorum sensing in Staphylococcus infections. J Clin Invest 112: 1620-1625.
- Otto M (2004) Quorum-sensing control in Staphylococci -- a target for antimicrobial drug therapy? FEMS Microbiol Lett 241: 135-141.
- Hirakawa H, Tomita H (2013) Interference of bacterial cell-to-cell communication: a new concept of antimicrobial chemotherapy breaks antibiotic resistance. Front Microbiol 4: 114.
- Jiang T, Li M (2013) Quorum sensing inhibitors: a patent review. Expert Opin Ther Pat 23: 867-894.
- de Nys R, Wright AD, Konig GM, Sticher O (1993) New halogenated furanones from the marine alga Delisea pluchra (cf. finbrata). Tetrahedron 49: 11213-11220.
- Zhu H, Kumar A, Ozkan J, Bandara R, Ding A, et al. (2008) Fimbrolide-coated antimicrobial lenses: their in vitro and in vivo effects. Optom Vis Sci 85: 292-300.
- Baveja JK, Willcox MD, Hume EB, Kumar N, Odell R, et al. (2004) Furanones as potential anti-bacterial coatings on biomaterials. Biomaterials 25: 5003-5012.
- Hume EB, Baveja J, Muir B, Schubert TL, Kumar N, et al. (2004) The control of Staphylococcus epidermidis biofilm formation and in vivo infection rates by covalently bound furanones. Biomaterials 25: 5023-5030.
- Wu H, Song Z, Hentzer M, Andersen JB, Molin S, et al. (2004) Synthetic furanones inhibit quorum-sensing and enhance bacterial clearance in Pseudomonas aeruginosa lung infection in mice. J Antimicrob Chemother 53: 1054-1061.
- Choi SC, Zhang C, Moon S, Oh YS (2014) Inhibitory effects of 4-hydroxy-2,5-dimethyl-3(2H)-furanone (HDMF) on acyl-homoserine lactone-mediated virulence factor production and biofilm formation in Pseudomonas aeruginosa PAO1. J Microbiol 52: 734-742.
- Hentzer M, Wu H, Andersen JB, Riedel K, Rasmussen TB, et al. (2003) Attenuation of Pseudomonas aeruginosa virulence by quorum sensing inhibitors. EMBO J 22: 3803-3815.
- Manefield M, Rasmussen TB, Henzter M, Andersen JB, Steinberg P, et al. (2002) Halogenated furanones inhibit quorum sensing through accelerated LuxR turnover. Microbiology 148: 1119-1127.
- Manefield M, Turner SL (2002) Quorum sensing in context: out of molecular biology and into microbial ecology. Microbiology 148: 3762-3764.
- Ren D, Bedzyk LA, Ye RW, Thomas SM, Wood TK (2004) Differential gene expression shows natural brominated furanones interfere with the autoinducer-2 bacterial signaling system of Escherichia coli. Biotechnol Bioeng 88: 630-642.
- Ren D, Sims JJ, Wood TK (2002) Inhibition of biofilm formation and swarming of Bacillus subtilis by (5Z)-4-bromo-5-(bromomethylene)-3-butyl-2(5H)-furanone. Lett Appl Microbiol 34: 293-299.
- Qazi S, Middleton B, Muharram SH, Cockayne A, Hill P, et al. (2006) N-acylhomoserine lactones antagonize virulence gene expression and quorum sensing in Staphylococcus aureus. Infect Immun 74: 910-919.
- Hume EB, Cole N, Khan S, Garthwaite LL, Aliwarga Y, et al. ( 2005) A Staphylococcus aureus mouse keratitis topical infection model: cytokine balance in different strains of mice. Immunol Cell Biol 83: 294-300.
- Manny AJ, Kjelleberg S, Kumar N, de Nys R, Read RW, et al. (1997) Reinvestigation of the sulfuric acid-catalysed cyclisation of brominated 2-alkyllevulinic acids to 3-alkyl-5-methylene-2(5H)-furanones. Tetrahedron 53: 15813-15826.
- Read R, Kumar N (2006) Inventors production of furanones patent US 7,064,220 B1.
- Oo TZ, Cole N, Garthwaite L, Willcox MD, Zhu H (2010) Evaluation of synergistic activity of bovine lactoferricin with antibiotics in corneal infection. J Antimicrob Chemother 65: 1243-1251.
- Odds FC (2003) Synergy, antagonism, and what the chequerboard puts between them. J Antimicrob Chemother 52: 1.
- Weeks AC, Balzli CL, Caballero A, Tang A, O'Callaghan R (2012) Identification and potency of cyclodextrin-lipid inhibitors of Staphylococcus aureus α-toxin. Curr Eye Res 37: 87-93.
- Garrett Q, Simmons PA, Xu S, Vehige J, Zhao Z, et al. (2007) Carboxymethylcellulose binds to human corneal epithelial cells and is a modulator of corneal epithelial wound healing. Invest Ophthalmol Vis Sci 48: 1559-1567.
- Xue ML, Zhu H, Willcox M, Wakefield D, Lloyd A, et al. (2001) The role of IL-1beta in the regulation of IL-8 and IL-6 in human corneal epithelial cells during Pseudomonas aeruginosa colonization. Curr Eye Res 23: 406-414.
- Dajcs JJ, Thibodeaux BA, Marquart ME, Girgis DO, Traidej M, et al. (2004) Effectiveness of ciprofloxacin, levofloxacin, or moxifloxacin for treatment of experimental Staphylococcus aureus keratitis. Antimicrob Agents Chemother 48: 1948-1952.
- Cole N, Bao S, Stapleton F, Thakur A, Husband AJ, et al. (2003) Pseudomonas aeruginosa keratitis in IL-6-deficient mice. Int Arch Allergy Immunol 130: 165-172.
- Kjelleberg S, Steinberg PD, Holmstrom C, Back A (1999) Inventors inhibition of gram positive bacteria.
- Jones MB, Jani R, Ren D, Wood TK, Blaser MJ (2005) Inhibition of Bacillus anthracis growth and virulence-gene expression by inhibitors of quorum-sensing. J Infect Dis 191: 1881-1888.
- Lattmann E, Sattayasai N, Schwalbe CS, Niamsanit S, Billington DC, et al. (2006) Novel anti-bacterials against MRSA: synthesis of focussed combinatorial libraries of tri-substituted 2(5H)-furanones. Curr Drug Discov Technol 3: 125-134.
- Parmar P, Salman A, Kalavathy CM, Kaliamurthy J, Prasanth DA, et al. (2006) Comparison of topical gatifloxacin 0.3% and ciprofloxacin 0.3% for the treatment of bacterial keratitis. Am J Ophthalmol 141: 282-286.
- Miller D (2013) Pharmacological treatment for infectious corneal ulcers. Expert Opin Pharmacother 14: 543-560.
- Mc Cormick CC, Caballero AR, Balzli CL, Tang A, O'Callaghan RJ (2009) Chemical inhibition of alpha-toxin, a key corneal virulence factor of Staphylococcus aureus. Invest Ophthalmol Vis Sci 50: 2848-2854.
- Chhabra SR, Harty C, Hooi DS, Daykin M, Williams P, et al. (2003) Synthetic analogues of the bacterial signal (quorum sensing) molecule N-(3-oxododecanoyl)-L-homoserine lactone as immune modulators. J Med Chem 46: 97-104.
- Jadhav GP, Chhabra SR, Telford G, Hooi DS, Righetti K, et al. (2011) Immunosuppressive but non-LasR-inducing analogues of the Pseudomonas aeruginosa quorum-sensing molecule N-(3-oxododecanoyl)-l-homoserine lactone. J Med Chem 54: 3348-3359.
- Hazlett LD (2004) Corneal response to Pseudomonas aeruginosa infection. Prog Retin Eye Res 23: 1-30.
- Xue ML, Thakur A, Cole N, Lloyd A, Stapleton F, et al. (2007) A critical role for CCL2 and CCL3 chemokines in the regulation of polymorphonuclear neutrophils recruitment during corneal infection in mice. Immunol Cell Biol 85: 525-531.